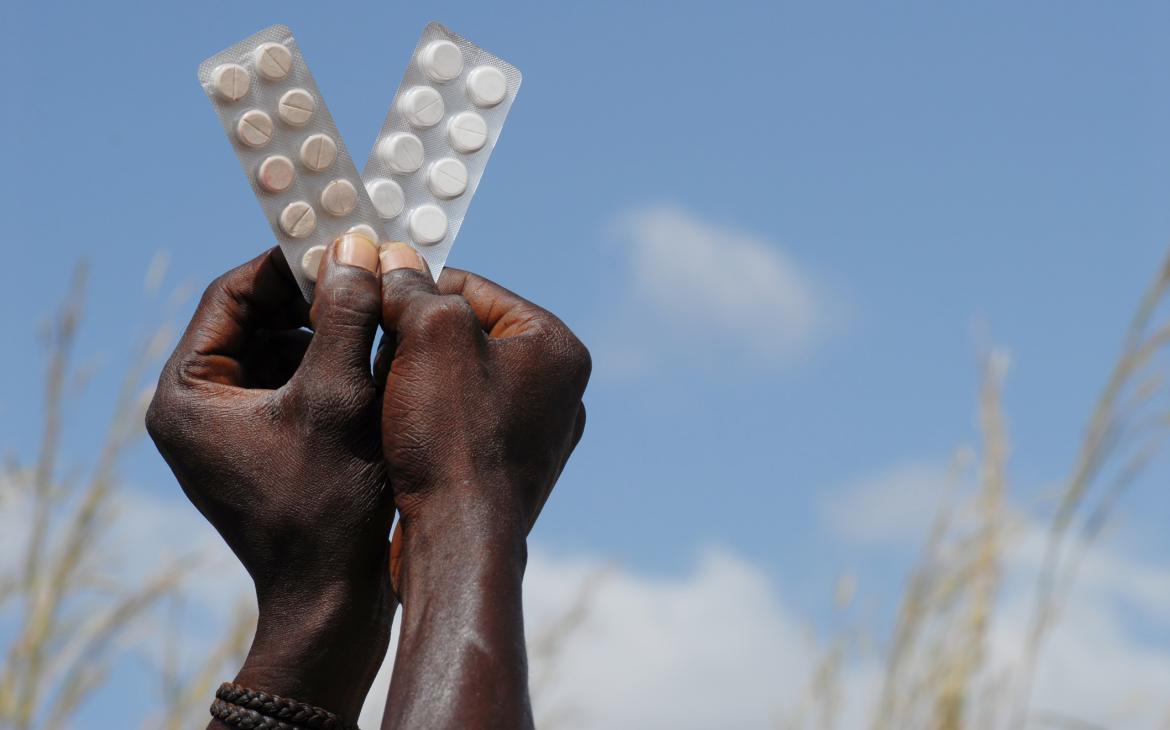
Quality Assurance is Key to Ending Malaria—For Good...
On this World Malaria Day, the world is calling for an end to malaria. We must increase efforts to ensure that antimalarials used to treat malaria are safe, effective and of good-quality. Here are some ways we can close the treatment gap and provide the millions of people newly infected with malaria each year with reliable, quality-assured medicines.








 Show 11 More
Show 11 More